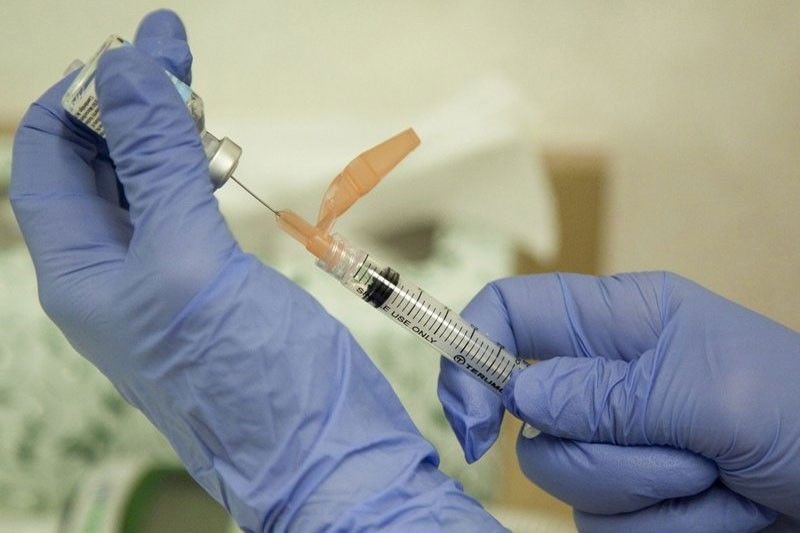
Koronadal City, South Cotabato – The Integrated Provincial Health Office (IPHO) of South Cotabato will conduct its “Ligtas Tigdas” Measles-Rubella Supplemental Immunization Activity (MR SIA) from October 26 to November 25, 2020 in designated places in the province.
In a press conference on October 13, 2020 by the IPHO represented by the National Immunization Program Coordinator Ms. Genesis Q. Navales, RN and IPHO Health Education and Information Officer II Ms. Hannah Ebeo, the vaccine is the only protection to children from measles, since there has been no cure or treatment for the disease yet.
German measles, also known as rubella, is a viral infection that causes a red rash on the body. Aside from the rash, people with German measles usually have a fever and swollen lymph nodes. The infection can spread from person to person through contact with droplets from an infected person’s sneeze or cough. This means that one can get German measles if he touches his mouth, nose, or eyes after touching something that has droplets from an infected person on it. One may also get German measles by sharing food or drinks with someone who’s infected.
Children should get two (2) doses of the vaccine, starting with the first dose at twelve (12) to fifteen (15) months of age, and the second dose at four (4) through six (6) years of age. The vaccine is given later than some other childhood vaccines because antibodies transferred from the mother to the baby can provide some protection from disease and make the vaccine less effective until about one (1) year old.
The IPHO urges the active cooperation of the parents and guardians for the MR SIA by voluntarily submitting their child/children to health workers in their community to have their shot of measles vaccine. Due to the current pandemic however, the vaccination will be given by strict schedules only, in order to avoid mass gatherings and the transmission of COVID-19. Also, no house-to-house immunization will transpire, considering the limited workforce in the province nowadays.
Parents and guardians are highly encouraged to inquire about the schedule of their children’s vaccination at their respective barangay health centers, barangay health stations, and the provincial hospital.
The success of this activity will benefit the children and community by creating a herd immunity which may prevent further transmission of highly infectious measles disease. It can be recalled that the entire immunization program of the national government through the Department of Health (DOH) has been adversely affected by the controversy brought by the Dengvaxia scare which caused panic among parents due to the misinformation and fake news.
Further, the health officials have assured the safety of the MR vaccine, since the vaccines that will be given during the MR SIA were already used decades ago and were already proven for their efficacy. There will only be some slight discomfort on the part of the child/children, since the method of administering the vaccine is through a subcutaneous injection, in other words, under the skin.
In this type of injection, a short needle is used to inject a drug into the tissue layer between the skin and the muscle. Medication given this way is usually absorbed more slowly than if injected into a vein, sometimes over a period of twenty four (24) hours. This type of injection is used when other methods of administration might be less effective.
Additionally, parents should not worry when their child/children would experience mild fever after the immunization, since fever is a common clinical complaint in adults and children with a variety of infectious illnesses, as well as a frequently reported adverse event following immunization.
According to the Infectious Diseases Society of America (IDSA), although the level of measured temperature indicative of a “fever” was first defined in 1868, it remains unclear what role fever has as a physiologic reaction to invading substances, how best to measure body temperature and compare measurements from different body sites, and, consequently, how to interpret fever data derived from vaccine safety trials or immunization safety surveillance. However, even with many aspects of the societal, medical, economic, and epidemiologic meanings of fever as an adverse event following immunization (AEFI) still elusive, it is a generally benign—albeit common—clinical sign.
The IPHO also assured the general public that only the health care providers such as registered nurses, medical technologists, and pharmacists, are authorized to administer vaccines, and these medical professionals are wearing the proper personal protective equipment (PPEs) during the conduct of the immunization. This is not just to protect themselves from COVID-19, but also to protect the children who will receive the vaccines.